Amrita AEEE syllabus pattern and evaluation
Amrita AEEE syllabus pattern and evaluation – The Amrita University Engineering Entrance examination for candidates to secure seats in the Amrita university need to follow the syllabus for the entrance test that has three subjects of Mathematics,Physics and Chemistry.The marking scheme as well as the breakup of marks for al three subjects is given below in the table.
Amrita AEEE syllabus pattern and evaluation
AEEE Pattern and Evaluation
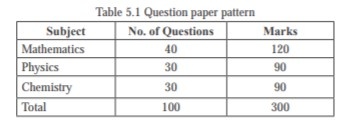
Amrita Entrance Examination–Engineering 2018 will be of 2 ½ hours duration and there will be one question paper
containing 100 objective type (multiple choice types) questions in Mathematics, Physics and Chemistry. Each question
will be followed by four answers of which one is correct/most appropriate.
Three marks will be awarded for every correct answer. For every incorrect answer, one mark will be deducted
from the total score. If no response is indicated in the answer sheet against a question, no marks, positive or negative will
be awarded. If more than one answer is indicated against a question, it will be treated as “incorrect answer” and negative
mark will be awarded. Candidates are advised to mark an answer only if they are sure that it is correct/the most appropriate.
Request for revaluation, re-checking or re-totaling will not be entertained.
SYLLABUS FOR ENTRANCE EXAMINATION
(See university website for model questions )
MATHEMATICS
a) COMPLEX NUMBERS
Complex numbers in the form a+ib and their representation in a plane. Argand diagram. Algebra of complex numbers,
Modulus and argument (or amplitude) of a complex number, square root of a complex number. Cube roots of unity,
triangle inequality.
b) PERMUTATIONS AND COMBINATIONS
Fundamental principle of counting; Permutation as an arrangement and combination as selection, Meaning of P(n,r)and
C(n, r).Simple applications.
c) BINOMIAL THEOREM
Binomial theorem for positive integral indices. Pascal’s triangle. General and middle terms in binomial expansions,
simple applications.
d) SEQUENCES AND SERIES
Arithmetic, Geometric and Harmonic progressions. Insertion of Arithmetic, Geometric and Harmonic means between
two given numbers. Relation between A.M., G.M. and H.M. Special series ?n, ?n2
, ?n3
. Arithmetico-Geometric
Series, Exponential and Logarithmic Series.
e) MATRICES AND DETERMINANTS
Determinants and matrices of order two and three, properties of determinants. Evaluation of determinants. Addition
and multiplication of matrices, adjoint and inverse of matrix. Solution of simultaneous linear equations using
determinants .
f) QUADRATIC EQUATIONS
Quadratic equations in real and complex number system and their solutions. Relation between roots and co-efficients,
nature of roots, formation of quadratic equations with given roots.
g) TRIGONOMETRY
Trigonometrical identities and equations. Inverse trigonometric functions and their properties. Properties of triangles,
including centroid, incentre, circumcentre and orthocentre, solution of triangles. Heights and distances.
h) MEASURES OF CENTRAL TENDENCY AND DISPERSION
Calculation of Mean, Median and Mode of grouped and ungrouped data. Calculation of standard deviation, variance
and mean deviation for grouped and ungrouped data.
i) PROBABILITY
Probability of an event, addition and multiplication theorems of probability and their applications; Conditional
probability; Bayes’ theorem, Probability distribution of a random variate; Binomial and Poisson distributions and
their properties.
j) DIFFERENTIAL CALCULUS
Polynomials, rational, trigonometric, logarithmic and exponential functions. Graphs of simple functions. Limits,
Continuity; differentiation of the sum, difference, product and quotient of two functions. Differentiation of
trigonometric, inverse trigonometric, logarithmic, exponential, composite and implicit functions; derivatives of order
upto two. Applications of derivatives: Maxima and Minima of functions one variable, tangents and normals, Rolle’s
and Langrage’s Mean Value Theorems.
k) INTEGRAL CALCULUS
Integral as an anti derivative. Fundamental integrals involving algebraic, trigonometric, exponential and logarithmic
functions. Integration by substitution, by parts and by partial fractions. Integration using trigonometric identities.
Integral as a limit of sum. Properties of definite integrals. Evaluation of definite integral; Determining areas of the
regions bounded by simple curves.
Information Handbook 27
Amrita Entrance Examination-Engineering 2018
l) DIFFERENTIAL EQUATIONS
Ordinary differential equations, their order and degree. Formation of differential equation. Solutions of differential equations
by the method of separation of variables. Solution of Homogeneous and linear differential equations, and those of type
d2
y/dx2
= f(x).
m) TWO DIMENSIONAL GEOMETRY
Review of Cartesian system of rectangular co-ordinates in a plane, distance formula, area of triangle, condition for the
collinearity of three points, slope of a line, parallel and perpendicular lines, intercepts of a line on the coordinate axes.
n) THE STRAIGHT LINE AND PAIR OF STRAIGHT LINES
Various forms of equations of a line, intersection of lines, angles between two lines, conditions for concurrence of
three lines, distance of a point from a line. Equations of internal and external bisectors of angles between two lines,
equation of family lines passing through the point of intersection of two lines, homogeneous equation of second degree
in x and y, angle between pair of lines through the origin, combined equation of the bisectors of the angles between
a pair of lines, condition for the general second degree equation to represent a pair of lines, point of intersections and
angles between two lines.
o) CIRCLES AND FAMILY OF CIRCLES
Standard form of equation of a circle, general form of the equation of a circle, its radius and centre, equation of a circle
in the parametric form, equation of a circle when the end points of a diameter are given, points of intersection of a line
and circle with the centre at the origin and condition for a line to be tangent, equation of a family of circles through
the intersection of two circles, condition for two intersecting circles to be orthogonal.
p) CONIC SECTIONS
Sections of cones, equations of conic sections ( parabola, ellipse and hyperbola) in standard forms, conditions for
y = mx+c to be a tangent and point(s) of tangency.
q) VECTOR ALGEBRA
Vector and scalars, addition of two vectors, components of a vector in two dimensional and three dimensional space,
scalar and vector products, scalar and vector triple product. Application of vectors to plane geometry.
r) THREE DIMENSIONAL GEOMETRY
Distance between two points. Direction cosines of a line joining two points. Cartesian and vector equation of a line.
Coplanar and skew lines. Shortest distance between two lines.Cartesian and vector equation of a plane. Angle between
(i) two lines (ii) two planes (iii) a line and a plane Distance of a point from a plane.
PHYSICS
a) UNITS AND DIMENSIONS
Units for measurement, system of units, SI, fundamental and derived units, dimensions and their applications.
b) MECHANICS
Motion in straight line, uniform and non-uniform motion, uniformly accelerated motion and its applications, Scalars
and Vectors, and their properties; resolution of vectors, scalar and vector products; uniform circular motion and its
applications, projectile motion Newton’s Laws of motion; conservation of linear momentum and its applications, laws
of friction, concept of work, energy and power; energy-kinetic and potential; conservation of energy; different forms
of energy. Elastic collisions in one and two dimensions.
Center of mass of a many particle system; center of mass of a rigid body, rotational motion and torque. Angular
momentum and its conservation. Moments of inertia, parallel and perpendicular axes theorem, moment of inertia for
a thin rod, ring, disc and sphere.
Gravitation: Acceleration due to gravity and its properties. One and two dimensional motion under gravity. Universal
law of gravitation, planetary motion, Kepler’s laws, artificial satellite-geostationary satellite, gravitational potential
energy near the surface of earth, gravitational potential and escape velocity.
28 Information Handbook
Amrita Entrance Examination-Engineering 2018
c) SOLIDS AND FLUIDS
Solids: Elastic properties, Hooke’s law, Young’s modulus, bulk modulus, modulus of rigidity. Liquids: Cohesion
and adhesion; surface energy and surface tension; flow of fluids, Bernoulli’s theorem and its applications; viscosity,
Stoke’s Law, terminal velocity.
d) OSCILLATIONS AND WAVES
Periodic motion, simple harmonic motion and its equation, oscillations of a spring and simple pendulum.
Wave motion, properties of waves, longitudinal and transverse waves, superposition of waves, Progressive and
standing waves. Free and forced oscillations, resonance, vibration of strings and air columns, beats, Doppler effect.
e) HEAT AND THERMODYNAMICS
Thermal expansion of solids, liquids and gases and their specific heats, relationship between Cp and Cv for gases,
first and second laws of thermodynamics, Carnot cycle, efficiency of heat engines. Transference of heat; thermal
conductivity; black body radiations, Kirchoff’s law, Wein’s Law, Stefan’s law of radiation and Newton’s law of cooling.
f) ELECTROSTATICS, CURRENT ELECTRICITY AND MAGNETOSTATICS
Coloumb’s law, dielectric constant, electric field, lines of force, field due to dipole, electric flux, Gauss’s theorem and
its applications; electric potential, potential due to a point charge; conductors and insulators, distribution of charge on
conductors; capacitance, parallel plate capacitor, combination of capacitors, energy stored in a capacitor.
Electric current: Cells-primary and secondary, grouping of cells; resistance and specific resistivity and its temperature
dependence. Ohm’s law, Kirchoff’s Law. Series and parallel circuits; Wheatstone’s Bridge and potentiometer with
their applications.
Heating effects of current, electric power, concept of thermoelectricity-Seebeck effect and thermocouple; chemical
effect of current- Faraday’s laws of electrolysis.
Magnetic effects: Oersted’s experiment, Biot Savert’s law, magnetic field due to straight wire, circular loop and
solenoid, force on a moving charge in a uniform magnetic field (Lorentz force), forces and torques on a current carrying
conductor in a magnetic field, force between current carrying wires, moving coil galvanometer and conversion to
ammeter and voltmeter.
Magnetostatics: Bar magnet, magnetic field, lines of force, torque on a bar magnet in a magnetic field, earth’s magnetic
field; para, dia and ferro magnetism, magnetic induction, magnetic susceptibility.
g) ELECTROMAGNETIC INDUCTION AND ELECTROMAGNETIC WAVES
Induced e.m.f., Faraday’s law, Lenz’s law, self and mutual inductance; alternating currents, impedance and
reactance, power in ac; circuits with L C and R series combination, resonant circuits, transformer and AC generator.
Electromagnetic waves and their characteristics; electromagnetic spectrum from gamma to radio waves.
h) RAY AND WAVE OPTICS
Reflection and refraction of light at plane and curved surfaces, total internal reflection; optical fiber; deviation and
dispersion of light by a prism; lens formula, magnification and resolving power; microscope and telescope, Wave
nature of light, interference, Young’s double experiment; thin films, Newton’s rings. Diffraction: diffraction due to a
single slit; diffraction grating, polarization and applications.
i) MODERN PHYSICS
Dual nature of Radiation – De Broglie relation, photoelectric effect, Alpha particle scattering experiment, atomic
masses, size of the nucleus; radioactivity, alpha, beta and gamma particles/rays. Radioactive decay law, half life and
mean life of radio active nuclei; Nuclear binding energy, mass energy relationship, nuclear fission and nuclear fusion.
Energy bands in solids, conductors, insulators and semiconductors, pn junction, diode, diode as a rectifier, transistor
action, transistor as an amplifier.
Information Handbook 29
Amrita Entrance Examination-Engineering 2018
CHEMISTRY
a) BASIC CONCEPTS
Atomic and molecular masses, mole concept and molar mass, percentage composition, empirical and molecular
formula, chemical reactions, stoichiometry and calculations based on stoichiometry.
b) ATOMIC STRUCTURE, CHEMICAL BONDING AND MOLECULAR STRUCTURE
Bohr’s model, de Broglie’s and Heisenberg’s principles, Quantum mechanical model, Orbital concept and filling
up of electrons; Bond formation and bond parameters; Valence bond and molecular orbital theory; VSEPR theory;
Hybridization involving s, p and d orbital; Hydrogen bond.
c) EQUILIBRIUM AND THERMODYNAMICS
Law of chemical equilibrium and Equilibrium Constant; Homogeneous and Heterogeneous equilibria; LeChatelier’s
principle, Ionic equilibrium; Acids, Bases, Salts and Buffers; Solubility product; Thermodynamic state; Enthalpy,
Entropy and Gibb’s free energy; Heats of reactions; Spontaneous and nonspontaneous processes.
d) ELECTROCHEMISTRY, KINETICS AND SURFACE CHEMISTRY
Specific, molar and equivalent conductance of weak and strong electrolytes; Kohlrausch law; Electrochemical cells
and Nernst equation; batteries, fuel cells and corrosion.
Rate of a reaction and factors affecting the rate: Rate constant, order and molecularity, collision theory.
Physisorption and chemisorptions; colloids and emulsions; homogeneous and heterogeneous catalysis.
e) SOLID STATE AND SOLUTIONS
Molecular, ionic, covalent and metallic solids; amorphous and crystalline solids; crystal lattices and Unit cells; packing
efficiency and imperfections; electrical and magnetic properties. Normality, molarity and molality of solutions, vapour
pressure of liquid solutions; ideal and non-ideal solutions, colligative proper-ties; abnormality.
f) HYDROGEN
Position of hydrogen in the periodic table; dihydrogen and hydrides- preparation and properties; water, hydrogen
peroxide and heavy water; hydrogen as a fuel.
g) S – BLOCK ELEMENTS
Group 1 and 2 Alkali and Alkaline earth elements; general characteristics of compounds of the elements; anomalous
behavior of the first element; preparation and properties of compounds like sodium and calcium carbonates, sodium
chloride, sodium hydroxide; biological importance of sodium, potassium and calcium.
h) P – BLOCK ELEMENTS
Groups 13 to 17 elements: General aspects like electronic configuration, occurrence, oxidation states, trends in
physical and chemical properties of all the families of elements; compounds of boron like borax, boron hydrides and
allotropes of carbon; compounds of nitrogen and phosphorus, oxygen and sulphur; oxides and oxyacids of halogens.
i) D, F – BLOCK ELEMENTS
Electronic configuration and general characteristics of transition metals; ionization enthalpy, ionic radii, oxidations
states and magnetic properties; interstitial compounds and alloy formation; lanthanides and actinoids and their
applications.
j) CO-ORDINATION COMPOUNDS
Werner’s theory and IUPAC nomenclature of coordination compounds; coordination number and isomerism; Bonding
in coordination compounds and metal carbonyls and stability; application in analytical methods, extraction of metals
and biological systems.
k) BASIC ORGANIC CHEMISTRY AND TECHNIQUES
Tetravalence of carbon and shapes or organic compounds; electronic displacement in a covalent bond inductive
and electromeric effects, resonance and hyperconjugation; hemolytic and heterolytic cleavage of covalent bond free
radicals, carbocations, carbanions electrophiles and nucleophiles; methods of purification of organic compounds;
qualitative and quantitative analysis.
30 Information Handbook
Amrita Entrance Examination-Engineering 2018
l) HYDROCARBONS, HALOALKANES AND HALOARENES
Alkanes, alkenes, alkynes and aromatic hydrocarbons; IUPAC nomenclature, isomerism; conformation of ethane,
geometric isomerism, general methods of preparation and properties, free radical mechanism of halogenations,
Markownikoff’s addition and peroxide effect; benzene, resonance and aromaticity, substitution reactions; Nature of
C-X bond in haloalkanes and haloarenes; mechanism of substitution reactions.
m) ALCOHOLS, PHENOLS AND ETHERS
IUPAC nomenclature, general methods of preparation, physical and chemical properties, identification of primary,
secondary and tertiary alcohols, mechanism of dehydration; electrophillic substitution reactions.
n) ALDEHYDES, KETONES, CARBOXYLIC ACIDS AND AMINES
Nomenclature, general methods of preparation, physical and chemical properties of the group members; nucleophilic
addition and its mechanism; reactivity of alpha hydrogen in aldehydes; mono and dicarboxylic acids-preparation and
reactions; identification of primary, secondary and tertiary amines; preparation and reactions of diazonium salts and
their importance in synthesis.
o) POLYMERS AND BIOMOLECULES
Natural and synthetic polymers, methods of polymerization, copolymerization, molecular weight of polymers,
Polymers of commercial importance, Carbohydrates: mono, oligo and polysaccharides; Proteins Alpha amino acid,
peptide linkage and polypeptides: Enzymes, Vitamins and Nucleic acids (DNA and RNA).
p) ENVIRONMENTAL CHEMISTRY
Air, water and soil pollution, chemical reactions in atmosphere, acid rain; ozone and its depletion; green house effect
and global warming; pollution control.
q) CHEMISTRY IN EVERYDAY LIFE
Drugs and their interaction; chemicals as analgesics, tranquilizers, antiseptics, antibiotics, antacids and antihistamines;
Chemicals in food- preservatives, artificial sweetening agents; cleansing agents – soaps and detergents.