CBSE syllabus for class 10 – Maths and Science
CBSE syllabus for class 10 – Maths and Science – This course curriculum or syllabus for the class 10th standard for the secondary school certificate examination that students have to study for the fresh exam as well as improvement / compartment exams.
Contents
CBSE syllabus for class 10 – Maths and Science
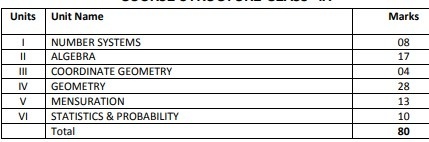
Breakup of marks for class 9 mathematics
Breakup of marks for class 10 mathematics
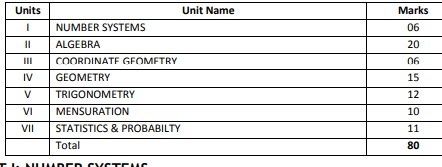
CBSE MATHS SYLLABUS FOR CLASS 10
UNIT I: NUMBER SYSTEMS
1. REAL NUMBER (15) Periods
Euclid’s division lemma, Fundamental Theorem of Arithmetic – statements after reviewing
work done earlier and after illustrating and motivating through examples, Proofs of
irrationality of ? ? ? Decimal representation of rational numbers in terms of
terminating/non-terminating recurring decimals.
UNIT II: ALGEBRA
1. POLYNOMIALS (7) Periods
Zeros of a polynomial. Relationship between zeros and coefficients of quadratic
polynomials. Statement and simple problems on division algorithm for polynomials with
real coefficients.
2. PAIR OF LINEAR EQUATIONS IN TWO VARIABLES (15) Periods
Pair of linear equations in two variables and graphical method of their
solution, consistency/inconsistency.
Algebraic conditions for number of solutions. Solution of a pair of linear equations in two
variables algebraically – by substitution, by elimination and by cross multiplication
method. Simple situational problems. Simple problems on equations reducible to linear
equations.
3. QUADRATIC EQUATIONS (15) Periods
Standard form of a quadratic equation ax2
+ bx + c = 0, (a ? 0). Solutions of quadratic
equations (only real roots) by factorization, by completing the square and by using quadratic
formula. Relationship between discriminant and nature of roots.
Situational problems based on quadratic equations related to day to day activities to be
incorporated.
4. ARITHMETIC PROGRESSIONS (8) Periods
Motivation for studying Arithmetic Progression Derivation of the nth term and sum of the
first n terms of A.P. and their application in solving daily life problems.
UNIT III: COORDINATE GEOMETRY
1. LINES (In two-dimensions) (14) Periods
Review: Concepts of coordinate geometry, graphs of linear equations. Distance formula.
Section formula (internal division). Area of a triangle.
UNIT IV: GEOMETRY
1. TRIANGLES (15) Periods
Definitions, examples, counter examples of similar triangles.
1. (Prove) If a line is drawn parallel to one side of a triangle to intersect the other two sides
in distinct points, the other two sides are divided in the same ratio.
2. (Motivate) If a line divides two sides of a triangle in the same ratio, the line is parallel to
the third side.
3. (Motivate) If in two triangles, the corresponding angles are equal, their corresponding sides
are proportional and the triangles are similar.
4. (Motivate) If the corresponding sides of two triangles are proportional, their corresponding
angles are equal and the two triangles are similar.
5. (Motivate) If one angle of a triangle is equal to one angle of another triangle and the sides
including these angles are proportional, the two triangles are similar.
6. (Motivate) If a perpendicular is drawn from the vertex of the right angle of a right triangle
to the hypotenuse, the triangles on each side of the perpendicular are similar to the whole
triangle and to each other.
7. (Prove) The ratio of the areas of two similar triangles is equal to the ratio of the squares of
their corresponding sides.
8. (Prove) In a right triangle, the square on the hypotenuse is equal to the sum of the squares
on the other two sides.
9. (Prove) In a triangle, if the square on one side is equal to sum of the squares on the other
two sides, the angles opposite to the first side is a right angle.
2. CIRCLES (8) Periods
Tangent to a circle at, point of contact
1. (Prove) The tangent at any point of a circle is perpendicular to the radius through the
point of contact.
2. (Prove) The lengths of tangents drawn from an external point to a circle are equal.
3. CONSTRUCTIONS (8) Periods
1. Division of a line segment in a given ratio (internally).
2. Tangents to a circle from a point outside it.
3. Construction of a triangle similar to a given triangle.
UNIT V: TRIGONOMETRY
1. INTRODUCTION TO TRIGONOMETRY (10) Periods
Trigonometric ratios of an acute angle of a right-angled triangle. Proof of their existence
(well defined); motivate the ratios whichever are defined at 0o and 90o. Values (with
proofs) of the trigonometric ratios of 300
, 450
and 600
. Relationships between the ratios.
2. TRIGONOMETRIC IDENTITIES (15) Periods
Proof and applications of the identity sin2
A + cos2
A = 1. Only simple identities to be given.
Trigonometric ratios of complementary angles.
3. HEIGHTS AND DISTANCES: Angle of elevation, Angle of Depression. (8) Periods
Simple problems on heights and distances. Problems should not involve more than two
right triangles. Angles of elevation / depression should be only 30°, 45°, 60°.
UNIT VI: MENSURATION
1. AREAS RELATED TO CIRCLES (12) Periods
Motivate the area of a circle; area of sectors and segments of a circle. Problems based on
areas and perimeter / circumference of the above said plane figures. (In calculating area
of segment of a circle, problems should be restricted to central angle of 60°, 90° and 120°
only. Plane figures involving triangles, simple quadrilaterals and circle should be taken.)
2. SURFACE AREAS AND VOLUMES (12) Periods
1. Surface areas and volumes of combinations of any two of the following: cubes, cuboids,
spheres, hemispheres and right circular cylinders/cones. Frustum of a cone.
2. Problems involving converting one type of metallic solid into another and other mixed
problems. (Problems with combination of not more than two different solids be taken).
UNIT VII: STATISTICS AND PROBABILITY
1. STATISTICS (18) Periods
Mean, median and mode of grouped data (bimodal situation to be avoided).
Cumulative frequency graph.
2. PROBABILITY (10) Periods
Classical definition of probability. Simple problems on single events (not using set
notation).
CBSE SCIENCE SYLLABUS FOR CLASS 10
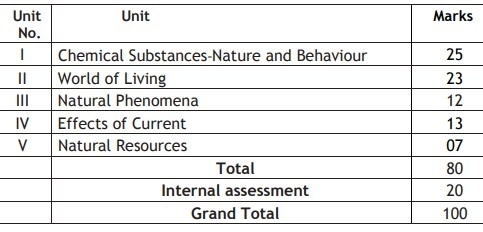
Theme: Materials (55 Periods)
Unit I: Chemical Substances – Nature and Behaviour
Chemical reactions: Chemical equation, Balanced chemical equation, implications of
a balanced chemical equation, types of chemical reactions: combination,
decomposition, displacement, double displacement, precipitation, neutralization,
oxidation and reduction.
Acids, bases and salts: Their definitions in terms of furnishing of H+ and OH– ions,
General properties, examples and uses, concept of pH scale (Definition relating to
logarithm not required), importance of pH in everyday life; preparation and uses of
Sodium Hydroxide, Bleaching powder, Baking soda, Washing soda and Plaster of Paris.
Metals and nonmetals: Properties of metals and non-metals; Reactivity series;
Formation and properties of ionic compounds; Basic metallurgical processes;
Corrosion and its prevention.
Carbon compounds: Covalent bonding in carbon compounds. Versatile nature of
carbon. Homologous series. Nomenclature of carbon compounds containing functional
groups (halogens, alcohol, ketones, aldehydes, alkanes and alkynes), difference
between saturated hydrocarbons and unsaturated hydrocarbons. Chemical properties of
carbon compounds (combustion, oxidation, addition and substitution reaction).
Ethanol and Ethanoic acid (only properties and uses), soaps and detergents.
Periodic classification of elements: Need for classification, Early attempts at
classification of elements (Dobereiner’s Triads, Newland’s Law of Octaves,
Mendeleev’s Periodic Table), Modern periodic table, gradation in properties, valency,
atomic number, metallic and non-metallic properties.
Theme: The World of the Living (50 Periods)
Unit II: World of Living
Life processes: ‘Living Being’. Basic concept of nutrition, respiration, transport and
excretion in plants and animals.
Control and co-ordination in animals and plants: Tropic movements in plants;
Introduction of plant hormones; Control and co-ordination in animals: Nervous
system; Voluntary, involuntary and reflex action; Chemical co-ordination: animal
hormones.
Reproduction: Reproduction in animals and plants (asexual and sexual) reproductive
health-need and methods of family planning. Safe sex vs HIV/AIDS. Child bearing and
women’s health.
Heredity and Evolution: Heredity; Mendel’s contribution- Laws for inheritance of
traits: Sex determination: brief introduction; Basic concepts of evolution.
Theme: Natural Phenomena (23 Periods)
Unit III: Natural Phenomena
Reflection of light by curved surfaces; Images formed by spherical mirrors, centre of
curvature, principal axis, principal focus, focal length, mirror formula (Derivation not
required), magnification.
Refraction; Laws of refraction, refractive index.
Refraction of light by spherical lens; Image formed by spherical lenses; Lens formula
(Derivation not required); Magnification. Power of a lens.
Functioning of a lens in human eye, defects of vision and their corrections,
applications of spherical mirrors and lenses.
Refraction oflightthrough a prism, dispersion oflight, scattering oflight, applications in
daily life.
Theme: How Things Work (32 Periods)
Unit IV: Effects of Current
Electric current, potential difference and electric current. Ohm’s law; Resistance,
Resistivity, Factors on which the resistance of a conductor depends. Series
combination of resistors, parallel combination of resistors and its applications in daily
life. Heating effect of electric current and its applications in daily life. Electric power,
Interrelation between P, V, I and R.
Magnetic effects of current : Magnetic field, field lines, field due to a current
carrying conductor, field due to current carrying coil or solenoid; Force on current
carrying conductor, Fleming’s Left Hand Rule, Electric Motor, Electromagnetic
induction. Induced potential difference, Induced current. Fleming’s Right Hand Rule,
Electric Generator, Direct current. Alternating current : frequency of AC. Advantage
of AC over DC. Domestic electric circuits.
Theme: Natural Resources (20 Periods)
Unit V: Natural Resources
Sources of energy: Different forms of energy, conventional and non-conventional
sources of energy: Fossil fuels, solar energy; biogas; wind, water and tidal energy;
Nuclear energy. Renewable versus non-renewable sources of Energy.
Our environment: Eco-system, Environmental problems, Ozone depletion, waste
production and their solutions. Biodegradable and non-biodegradable substances.
Management of natural resources: Conservation and judicious use of natural
resources. Forest and wild life; Coal and Petroleum conservation. Examples of
people’s participation for conservation of natural resources. Big dams: advantages
and limitations; alternatives, if any. Water harvesting. Sustainability of natural resources.