First year engineering semester 1 syllabus
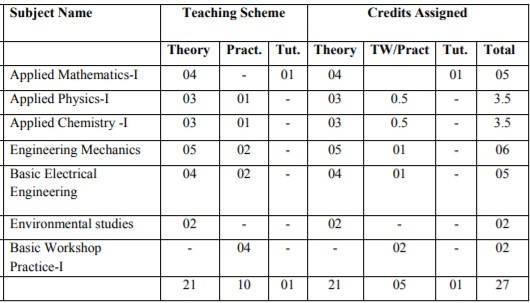
First year engineering semester 1 syllabus – The syllabus for the first year engineering F.E for Mumbai University Bachelors degree course has six main subjects and a practical workshop lab.This is an extremely hard year and being the first semester you need to work extra to clear these main subjects.Watch out for mechanics and Applied Maths 1 , they are absolute killers and if there is a high chance you will end up with ATKTs in F.E.
IMPORTANT
Finish study of hard subject FIRST, easy subjects can be completed in 1 week time
DO NOT under any circumstances, try and cover all the topics here ,leave out 25% in all subjects,and the rest 75% be a master of it. If you try and be a hero ,you will fail.
Contents
First year engineering semester 1 syllabus
APPLIED MATHEMATICS 1
Module?1: Complex Numbers
Pre?requisite: Review of Complex Numbers?Algebra of Complex Number,
Different representations of a Complex number and other definitions,
D’Moivre’s Theorem.
1.1.Powers and Roots of Exponential and Trigonometric Functions.
1.2. Expansion of sinn ?, cosn ? in terms of sines and cosines of multiples
of ? and Expansion of sinn?, cosn? in powers of sin?, cos?
1.3.Circular functions of complex number and Hyperbolic functions.
Inverse Circular and Inverse Hyperbolic functions. Separation of real
and imaginary parts of all types of Functions.
3 hrs
2 hrs
4 hrs
2
Module?2:Logarithm of Complex Numbers , Successive Differentiation
2.1. Logarithmic functions, Separation of real and Imaginary parts of
Logarithmic Functions.
2.2. Successive differentiation: nth derivative of standard functions.
Leibnitz’s Theorem (without proof) and problems
4 hrs
4 hrs
3
Module?3:Matrices
Pre?requisite: Inverse of a matrix, addition, multiplication and transpose of a
matrix
3.1. Types of Matrices (symmetric, skew? symmetric, Hermitian, Skew
Hermitian, Unitary, Orthogonal Matrices and properties of Matrices). Rank
of a Matrix using Echelon forms, reduction to normal form, PAQ in normal
form, system of homogeneous and non –homogeneous equations, their
consistency and solutions. Linear dependent and independent
vectors. Application of inverse of a matrix to coding theory.
9 hrs
4
Module?4: Partial Differentiation
4.1. Partial Differentiation: Partial derivatives of first and higher order. Total
differentials, differentiation of composite and implicit functions.
4.2. Euler’s Theorem on Homogeneous functions with two and three
independent variables (with proof).Deductions from Euler’s Theorem
6 hrs
3 hrs
6
5
Module?5: Applications of Partial Differentiation , Expansion of
Functions
1.1 Maxima and Minima of a function of two independent variables,
Jacobian.
1.2 Taylor’s Theorem (Statement only) and Taylor’s series, Maclaurin’s
series (Statement only).Expansion of ???? , sin(x), cos(x), tan(x), sinh(x),
cosh(x), tanh(x), log(1+x), ???????1(??),???????1(??),???????1(??), Binomial
series.
4 hrs
4 hrs.
6
Module?6: Indeterminate forms, Numerical Solutions
of Transcendental Equations and System of Linear Equations
6.1. Indeterminate forms, L? Hospital Rule, problems involving
series.
6.2. Solution of Transcendental Equations: Solution by Newton Raphson
method and Regula –Falsi Equation.
6.3. Solution of system of linear algebraic equations, by (1) Gauss
Elimination Method, (2) Gauss Jacobi Iteration Method, (3) Gauss Seidal
Iteration Method. (Scilab programming for above methods is to be
taught during lecture hours)
APPLIED PHYSICS 1
CRYSTAL STRUCTURE
Introduction to crystallography; Study of characteristics of unit cell of
Diamond, ZnS, NaCl and HCP; Miller indices of crystallographic planes &
directions; interplanar spacing; X-ray diffraction and Bragg’s law;
Determination of Crystal structure using Bragg’s diffractometer; Frenkel and
Schotkey crystal defects; Ionic crystal legancy (3,4,6,8); Liquid crystal phases.
07 hrs
Module 2 QUANTUM MECHANICS
Introduction, Wave particle duality; de Broglie wavelength; experimental
verification of de Broglie theory; properties of matter waves; wave packet,
phase velocity and group velocity; Wave function; Physical interpretation of
09 hr
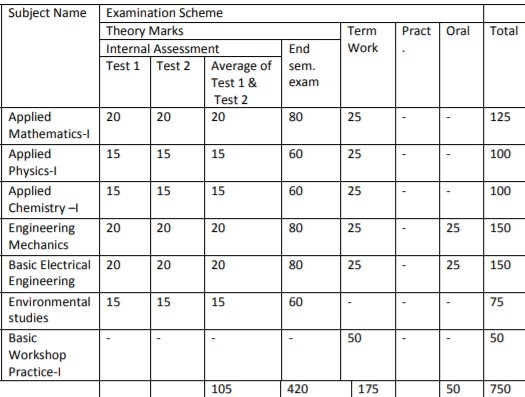
Subject
Code
Subject
Name
Examination Scheme
Theory
Term
Work Practical Oral Total Internal Assessment End SEM.
Exam. Test
1
Test
2
Average of
Test 1 & 2
FEC102 Applied
Physics-I
15 15 15 60 25 – – 100
9
wave function; Heisenberg’s uncertainty principle; Electron diffraction
experiment and Gama ray microscope experiment; Applications of uncertainty
principle; Schrodinger’s time dependent wave equation; time independent
wave equation; Motion of free particle; Particle trapped in one dimensional
infinite potential well.
Module 3 SEMICONDUCTOR PHYSICS
Splitting of energy levels for band formation; Classification of
semiconductors(direct & indirect band gap, elemental and compound);
Conductivity, mobility, current density (drift & diffusion) in semiconductors(n
type and p type); Fermi Dirac distribution function; Fermi energy level in
intrinsic & extrinsic semiconductors; effect of impurity concentration and
temperature on fermi level; Fermi Level diagram for p-n junction(unbiased,
forward bais, reverse bias); Breakdown mechanism (zener & avalanchy), Hall
Effect
Applications of semiconductors: Rectifier diode, LED, Zener diode, Photo
diode, Photovoltaic cell, BJT, FET, SCR., MOSFET
14 hrs
Module 4 SUPERCONDUCTIVITY
Introduction, Meissner Effect; Type I and Type II superconductors; BCS
Theory (concept of Cooper pair); Josephson effect
Applications of superconductors- SQUID, MAGLEV
03 hrs
Module 5 ACOUSTICS
Conditions of good acoustics; Reflection of sound(reverberation and echo);
absorption of sound; absorption coefficient; Sabine’s formula; Acoustic Design
of a hall; Common Acoustic defects and acoustic materials
03 hrs
Module 6 ULTRASONICS
Ultrasonic Wave generation; Magnetostriction Oscillator; Piezoelectric
Oscillator;
Applications of ultrasonic: Eco sounding; NDT; ultrasonic
cleaning(cavitation); ultrasonic sensors; Industrial applications of
ultrasonic(soldering, welding, cutting, drilling)
APPLIED CHEMISTRY 1
Water
Impurities in water, Hardness of water, Determination of Hardness of water by EDTA
method and problems, Softening of water by Hot and Cold lime Soda method and
numerical problems. Zeolite process and numerical problems. Ion Exchange process
and numerical problems. Potable water standard as per BIS w.r.t. i) pH, ii) Alkalinity,
iii) TDS, iv) Hardness; Drinking water or Municipal water -Treatments removal of
microorganisms by adding Bleaching powder, Chlorination (no breakpoint
chlorination), Disinfection by Ozone, Electrodialysis, Reverse osmosis, and Ultra
filtration. BOD, COD- definition & significance, sewage treatment (only activated
sludge process), Numerical problems related to COD.
12 hrs
Module 2 Polymers
Introduction to polymers, Classification, Types of polymerization, Thermoplastic and
Thermosetting plastic; Compounding of plastic,Fabrication of plastic by Compression,
Injection, Transfer and Extrusion moulding. Preparation, properties and uses of Phenol
formaldehyde, PMMA, Kevlar. Effect of heat on the polymers (Glass transition
temperature), Viscoelasticity. Conducting polymers, Engineering Plastics, Polymers in
medicine and surgery. Rubbers :
Natural rubber- latex, Drawbacks of natural rubber, Vulcanization of rubber,
12 hrs
Subject
Code Subject Name
Examination Scheme
Theory
Term
Work Practical Oral Total Internal Assessment End
SEM.
Exam.
Test
1
Test
2
Average of
Test 1 & 2
FEC103 Applied
Chemistry-I
15 15 15 60 25 – – 100
12
Preparation, properties and uses of Buna-S, Silicone and Polyurethane rubber.
Module 3 Lubricants
Introduction, Definition, Mechanism of lubrication, Classification of lubricants, Solid
lubricants (graphite & Molybdenum disulphide), Semisolid lubricants, Liquid
lubricants, Additives in blended Oils. Important properties of lubricants – Definition
and significance of – Viscosity, Viscosity index, Flash and fire points, Cloud and pour
points, Oiliness, Emulsification, Acid value and numerical problems, Saponification
value and numerical problems.
07 hrs
Module 4 Phase Rule
Gibb’s Phase Rule, Terms involved with examples, One Component System (Water),
Reduced Phase Rule, Two Component System (Pb- Ag), Advantages and Limitations
of Phase Rule.
04 hrs
Module 5 Important Engineering Materials
Cement – Manufacture of Portland Cement, Chemical Composition and Constitution of
Portland Cement, Setting and Hardening of Portland Cement, Concrete, RCC and
Decay.
Nanomaterials, preparation (Laser and CVD) method, properties and uses of CNTS,
Fullerene – properties and uses.
ENGINEERING MECHANICS
1.1 System of Coplanar Forces:
Resultant of concurrent forces, parallel forces, non-concurrent
non-parallel system of forces, Moment of force about a point, Couples, Varignon’s
Theorem. Force couple system. Distributed Forces in plane.
1.2 Centroid for plane Laminas.
05
04
15
02 2.1Equilibrium of System of Coplanar Forces:
Condition of equilibrium for concurrent forces, parallel forces and non-concurrent
non-parallel general forces and Couples.
2.2Types of support: Loads, Beams, Determination of reactions at supports for
various types of loads on beams.(Excluding problems on internal hinges)
2.3Analysis of plane trusses: By using Method of joints and Method of
sections.(Excluding pin jointed frames)
06
03
05
03 3.1 Forces in space:
Resultant of Non-coplanar Force Systems: Resultant of concurrent force system,
parallel force system and non-concurrent non-parallel force system.
Equilibrium of Non-coplanar Force Systems: Equilibrium of Concurrent force
system, parallel force system and non-concurrent non-parallel force system.
3.2 Friction:
Introduction to Laws of friction, Cone of friction, Equilibrium of bodies on inclined
plane, Application to problems involving wedges, ladders.
3.3 Principle of virtual work:
Applications on equilibrium mechanisms, pin jointed frames.
05
07
04
04 4.1 Kinematics of a Particle: -Rectilinear motion, Velocity & acceleration in terms
of rectangular co-ordinate system, Motion along plane curved path, Tangential&
Normal component of acceleration, Motion curves (a-t, v-t, s-t curves), Projectile
motion.
10
05 5.1 Kinematics of a Rigid Body :- Introduction to general plane motion,
Instantaneous center of rotation for the velocity, velocity diagrams for bodies in plane
motion.
06
06 6.1 Kinetics of a Particle: Force and Acceleration: -Introduction to basic concepts,
D’Alemberts Principle, Equations of dynamic equilibrium, Newton’s second law of
motion.
6.2 Kinetics of a Particle: Work and Energy: Principle of work and energy, Law of
conservation of energy.
6.3 Kinetics of a Particle: Impulse and Momentum: Principle of linear impulse and
momentum. Law of conservation of momentum. Impact and collision.
Basic Electrical engineering
DC Circuits(Only Independent Sources): Kirchhoff ’s laws, Ideal and
practical voltage and current source, Mesh and Nodal analysis, Supernode and
Supermesh analysis, Source transformation, Star-delta transformation,
Superposition theorem, Thevenin’s theorem, Norton’s theorem, Maximum
power transfer theorem, (Source transformation not allowed for Superposition
theorem, Mesh and Nodal analysis).
18
02
AC Circuits: Generation of alternating voltage and currents, RMS and Average
value, form factor, crest factor, AC through resistance, inductance and
capacitance, R-L, R-C and R-L-C series and parallel circuits, phasor diagrams,
power and power factor, series and parallel resonance, Q factor and bandwidth.
12
03
Three Phase Circuits: Three phase voltage and current generation, star and
delta connections(balanced load only), relationship between phase and line
currents and voltages, Phasor diagrams, Basic principle of wattmeter,
measurement of power by one and two wattmeter methods.
06
04
Single Phase Transformer: Construction, working principle, emf equation,
ideal and practical transformer, transformer on no load and on load, phasor
diagrams, equivalent circuit, OC and SC test, regulation and efficiency.
12
05 DC Machines: Principle of operation of DC motors and DC generators,
construction and classification of DC machines, emf equation.
ENVIRONMENTAL STUDIES
Overview of Environmental Aspects:
• Definition, Scope and Importance of Environmental Study
• Need for Public awareness of environmental education
• Introduction to depletion of natural resources: Soil, Water, Minerals and
Forests.
• Global crisis related to – Population, water, sanitation & Land.
Ecosystem:
• Study of ecosystems: Forest, desert and aquatic (in brief).
• Energy flow in Ecosystem, overview of Food Chain, Food Web and
Ecological Pyramid.
• Concept of ecological succession and its impact on human beings (in brief).
Case Study on Chipko Movement (Uttarakhand, India), (began in 1973).
4
Module 2 Aspects of Sustainable Development:
• Concept and Definition of Sustainable Development.
• Social, Economical and Environmental aspects of sustainable development.
• Control measures: 3R (Reuse, Recovery, Recycle),
• Resource utilization as per the carrying capacity (in brief).
Case Study on Narmada Bachao Andolan (Gujarat, India, in the mid and late
1980s).
2
Module 3 Types of Pollution:
• Water pollution: Sources of water pollution and Treatment of Domestic and
industrial waste water (with flow-diagram of the treatment),
• Land Pollution: Solid waste, Solid waste management by land filling,
8
21
composting and incineration
• Air pollution: Sources of air pollution,
Consequences of air pollution :-
Greenhouse effect (Explanation with schematic diagram),
Photochemical Smog (Explanation with chemical reaction).
Cleaning of gaseous effluents to reduce air contaminants namely dust
particle or particulate matters by using:- (i) Electrostatic precipitators (ii)
Venturi scrubber (Schematic diagram and working).
• Noise pollution: Sources, effects, threshold limit for different areas and
control methods.
• E-Pollution: Definition, Sources and effects.
• Nuclear pollution: Sources and effects.
Case study on Water Pollution of Ganga River.
Case study on London smog (U. K.)(December, 1952).
Case Study of Fukushima Disaster (March, 2011).
Module 4 Pollution Control Legislation:
• Functions and powers of Central and State Pollution Control Board.
• Environmental Clearance, Consent and Authorization Mechanism.
Case Study of Dombivali MIDC- Boiler Blast Tragedy (Thane, Maharashtra,
India), (May, 2016).
3
Module 5 Renewable Sources of Energy:
• Importance of renewable sources of energy.
• Principle and working with schematic diagram of :-
(i) Solar Energy: (a) Flat plate collector and (b) Photovoltaic cell.
(ii) Wind Energy: Wind Turbines.
(iii) Hydropower: Hydropower generation from water reservoir of the dam.
(iv) Geothermal Energy: Utilisation of underground sources of steam for
power generation.
4
Module 6 Technological Advances to overcome Environmental problems:
• Concept of Green Buildings,
• Various indoor air pollutants and their effects on health.
• Carbon Credit: Introduction and general concept.
• Disaster Management: Techniques of Disaster Management to cope up with
(i) Earthquake and (ii) Flood.
Case Study on Earthquake in Latur (Maharashtra, India), (September,1993).
Case Study on Cloudburst and Landslides at Kedarnath (Uttarakhand, India),
(June, 2013)
How to solve mech kt
do all 100% theory+derivations, numericals can be hard so avoid